This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What are post-translational modifications?
Post-translational modifications (PTMs) involve covalently adding functional groups to proteins, proteolytically cleaving regulatory subunits, or degrading entire proteins. There are multiple different post-translational modifications that can occur, including: glycosylation, phosphorylation, ubiquitination, nitrosylation, methylation, acetylation, lipidation, and proteolysis. These modifications can modify any aspect of a protein's function, therefore, these modifications are critical to understanding the regulation and activity of proteins. (1)
What is phosphorylation?
Phosphorylation is an important mechanism involved in regulating protein function and signaling within the cell. Phosphorylation usually occurs on either serine, threonine, or tyrosine residues of the protein. Phosphorylation normally occurs when a protein-kinase transfers a phosphate group from ATP to the amino acid residue on the protein of interest. Because phosphorylation is so important in cellular processes, phosphoproteomics, or the study of the phosphorylation of proteins, has been an important area of research.
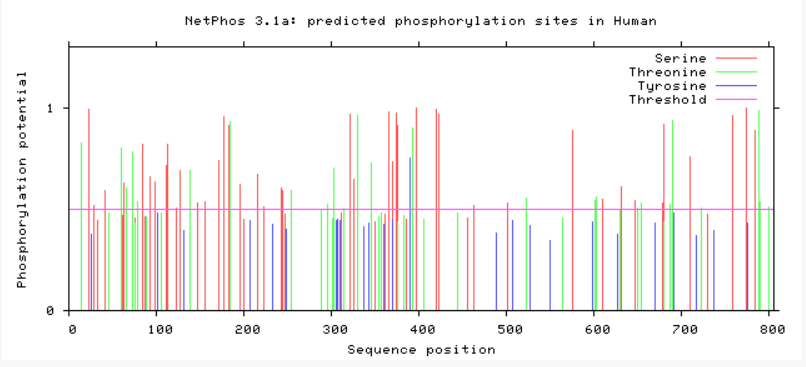
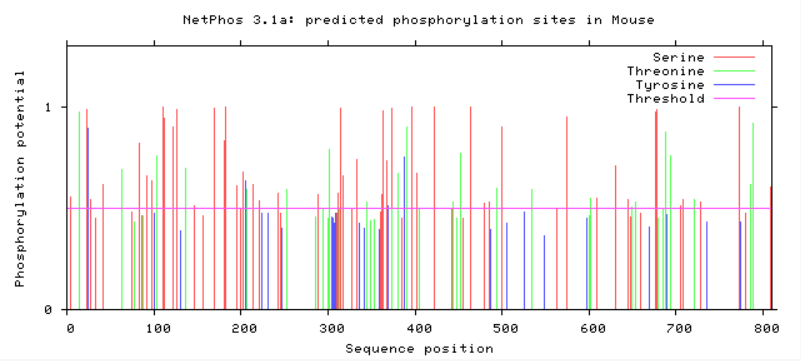
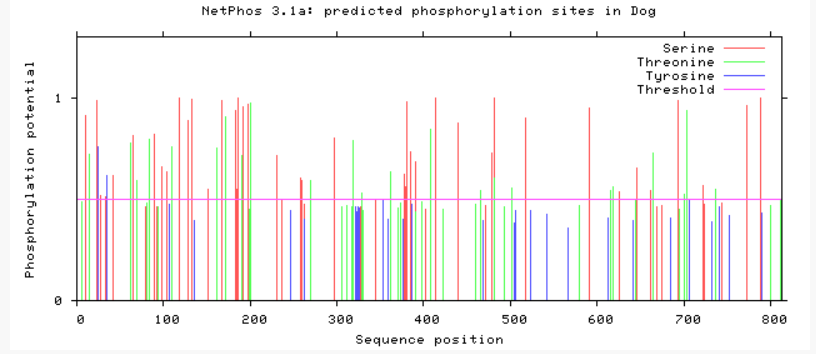
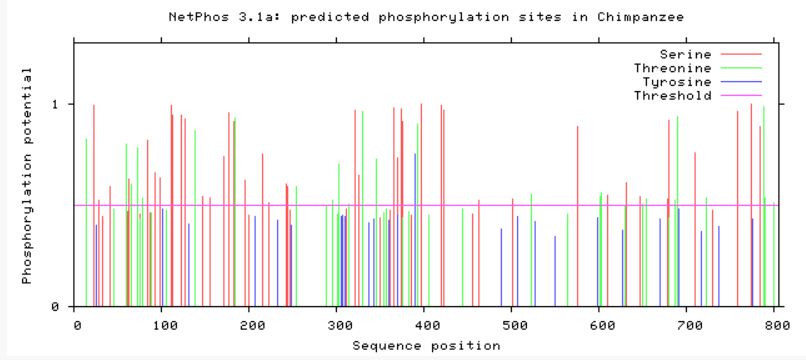
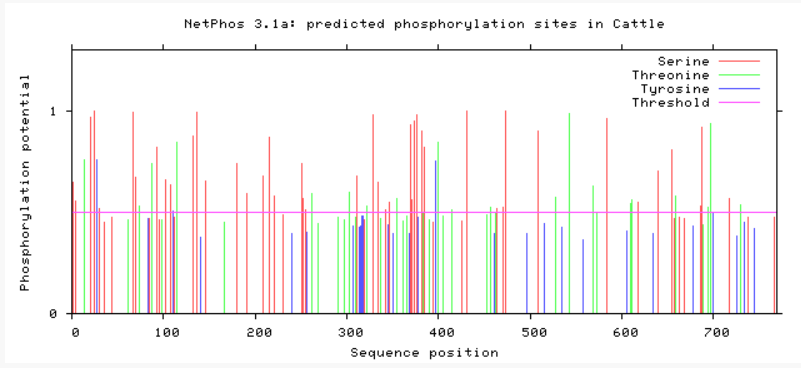
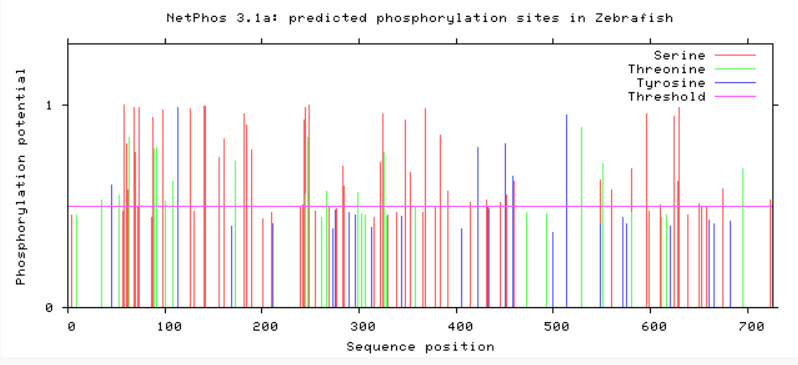
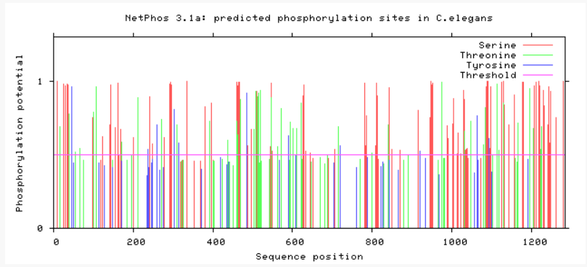
Are phosphorylation sites conserved between species?
Conclusion
As evident from above, TMC6 has a myriad of of different phosphorylation sites that could be involved in differing functions or regulation of TMC6. A conserved area in TMC6 in humans, mice, chimpanzees, and dogs is around 275, in which there is no phosphorylation at this spot. This conserved area is lost in cattle, zebrafish, and C. elegans. C. elegans shows a profound difference in phosphorylation among the residues. All in all, the phosphorylation sites in humans, mice, and chimpanzee are heavily conserved.
Sources:
1. https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
2. https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/phosphorylation.html
3. http://www.cbs.dtu.dk/services/NetPhos/
1. https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
2. https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/phosphorylation.html
3. http://www.cbs.dtu.dk/services/NetPhos/