This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What is a Protein Interaction Network?
Protein Interaction Networks (PINs) are mathematical representations of the protein-protein interactions (PPI) found within the cell. These are essential to understand the functions and physiology of normal cells and proteins. The connections between different proteins are highly specific and have particular biological functions. To find PPI, Yeast-two-hybrid screens or affinity purification can be used. To form the PINS, databases like String, IntAct, and BioGrid are used. These databases can be used for proteins such as TMC6 to form the protein network that interacts with TMC6. (1)
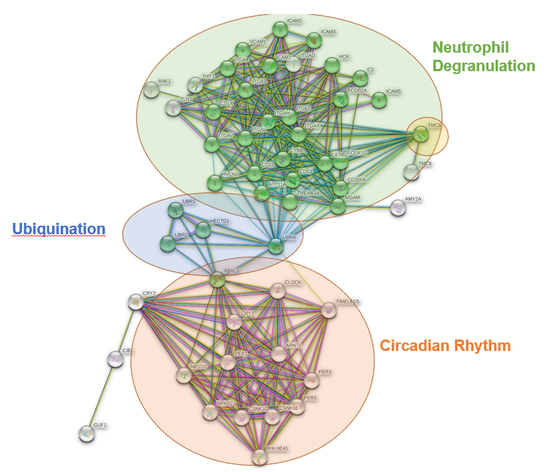
TMC6 Protein Interaction Network
|
TMC6 is a transmembrane channel protein that is known to colocalize and form a complex with TMC8 and CIB1. In the protein interaction network of TMC6 in Homo sapiens (left) it shows that TMC6 interacts with a plethora of proteins that have roles in neutrophil degranulation. Many of these proteins also play a role in integrin-binding. These proteins interact with multiple different proteins involved with circadian rhythm through ubiquinating-proteins. These three molecular processes show TMC6's advanced role in both innate immunity (neutrophils) and the development of squamous-cell carcinoma (circadian rhythm). |
H. sapiens (Humans) and M. musculus (Mice) TMC6 Interactions
|
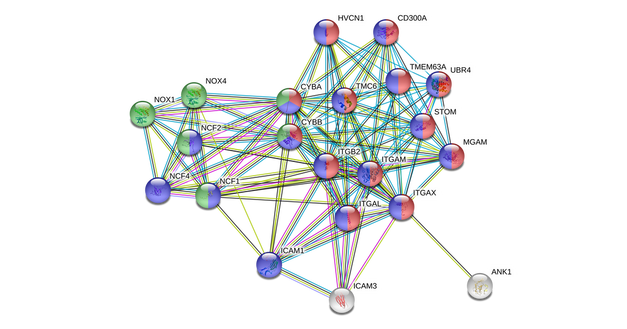
Homo sapiens (Humans)
Most of the proteins that directly link to TMC6 in humans are involved in neutrophil degranulation (red) and the immune response (blue). As the network expands to include indirect interactions, it is shown that TMC6 indirectly interacts with proteins that are involved in superoxide-generating NADPH oxidase activity (green). Multiple of these proteins, including NOX4, are involved in cell-signaling processes. |
|
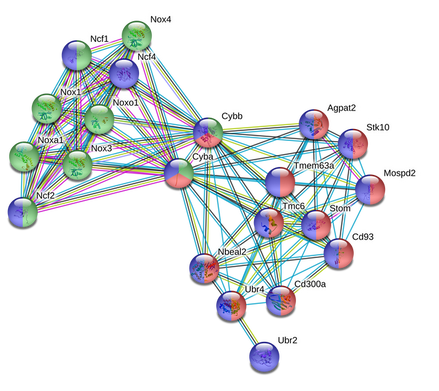
Mus musculus (Mice)
Most of the proteins that directly link to TMC6 in mice are involved in neutrophil degranulation (red) and the immune response (blue). As the network expands to include indirect interactions, it is shown that TMC6 indirectly interacts with proteins that are involved in superoxide-generating NADPH oxidase activity (green) through two "bridge proteins" in Cybb and Cyba. Both of these proteins are critical components of membrane-bound oxidase of phagocytes. Nox4 is also involved in cell-signaling processes. |
Conclusion
Both in humans and in mice TMC6 heavily interacts with proteins involved in neutrophil degranulation, immune response, and superoxide-generating NADPH oxidase activity. The first two processes are distinctly involved in EV, as a lack of cell-mediated immunity (given by neutrophils and immune response) leads to beta-HPV infection and the progression of EV. Interestingly TMC6 had interactions with oxidase activity proteins, which are involved in multiple different processes, including: cell-signaling, otolith formation, and host-cell defense mechanisms.