This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What is epidermodysplasia verruciformis?
Epidermodysplasia verruciformis (EV), also known as Lewandowski and Lutz Syndrome or "Tree Man" Syndrome, is a rare autosomal recessive skin disease that leads to a high susceptibility to infection by human papillomaviruse (HPV), specifically HPV 5 and 8. This infection leads to a high risk of skin carcinoma, persistent scaly wart-like and/or macular lesions, and possibly cutaneous horns. The typical form of EV is caused by biallelic null mutations in either TMC6 (EVER1) or TMC8 (EVER2). (3)
Disease Overview
Discovery and ProgressFelix Lewandowsky and Wilhelm Lutz discovered the skin condition known as EV in 1922. They classified it as an epidermal nevus, which is an abnormal, noncancerous patch of skin caused by an overgrowth of the epidermis. In 1939, Sullivan and Ellis found a significant link between EV and an increased risk of skin cancer. At this time it was reasoned that EV was transmitted via a recessive gene because most patients had consanguineous parents. Over time, it was discovered that EV is caused by persistent infection of cutaneous tissue by two strains of HPV. These two strains usually cause no symptoms in normal adults; however, in EV patients, because of a lack of cell-mediated immunity, they can lead to carcinomas. Over time it was discovered that typical EV presents because of mutations in genes EVER1 and/or EVER2. (3) |
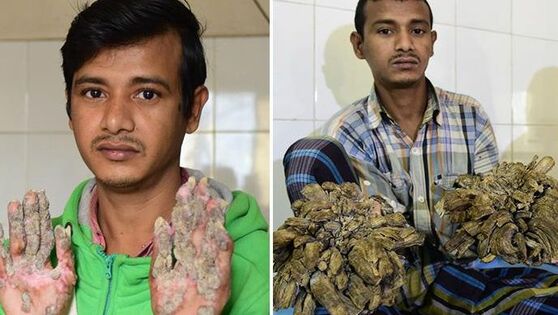
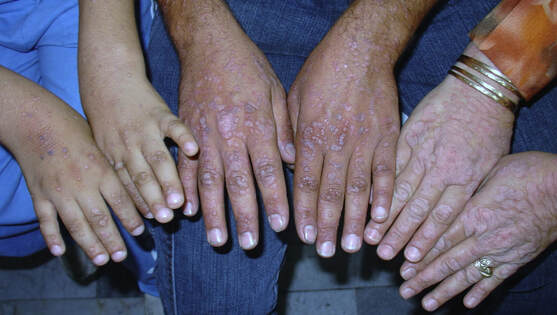
Symptoms OverviewEV patients normally present at a young age with progressive development of flat warts (shown above), hyperpigmented or hypopigmented plaques, seborrheic keratosis-like lesions and pityriasis versicolor-like lesions on areas that are exposed to the sun frequently. Due to infection by oncogenic HPV sub-types 5 and 8, up to 70% of patients develop non-melanoma skin cancer originating from the infected warts. These cancers are normally squamous cell carcinomas (SCC) and usually develop 10-30 years after the initial presentation of the disease in areas that get frequent sun exposure. The most dramatic symptom seen in EV patients are the keratotic warts, which can overgrow and create cutaneous horns, dramatically affecting everyday life (above, left). These outgrowths in themselves are benign, but their frequency is increased by SCC. (1) |
TMC6 (EVER1) Gene
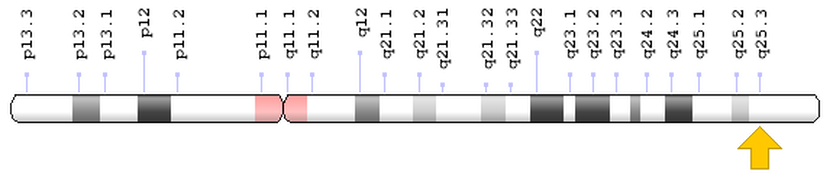
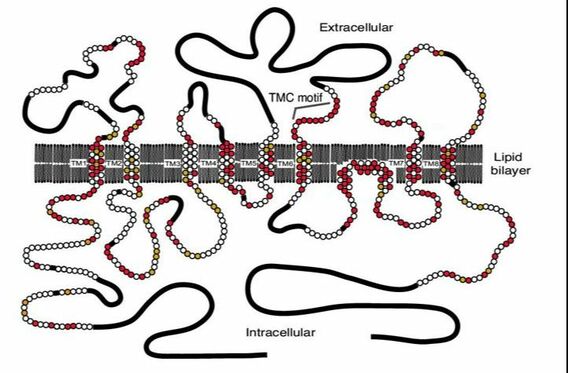
The TMC6 Gene, also known as EVER1, is located on chromosome 17q25.3, and is adjacent to TMC8 (EVER2). As of 2016, there are seven known mutations in the EVER1 gene that have contributed to EV. The EVER1 gene is made up of 20 exons and 19 introns and encodes four transcripts. All TMC proteins contain a 120-amino acid sequence, considered the TMC domain. TMC6 encodes an integral membrane protein, that although unknown, predictably forms a transmembrane channel that has 10 transmembrane domains, two leucine zipper motifs (2), and two putative glycosylation sites (4). The full length TMC6 gene produces two 805-amino acid sequences, and in alternative splicing events two smaller isoforms of 384 and 454-amino acids can be synthesized (4). These proteins form a complex with the Zinc transporter protein ZnT-1 in the endoplasmic reticulum (ER)(1), and are transcribed in keratinocytes, CD4+ and CD8+ lymphocytes, B lymphocytes, endothelial cells, and NK cells, with the highest expression in the lymphoid cells(4).
EVER1 Role In Persistent HPV
|
One of the main characteristics of EV is persistent HPV infections caused by defective cell-mediated immunity (3). Although it is unknown how EVER proteins affect and maintain HPV immunity, it is hypothesized that EVER1 and EVER2 resist HPV infection in healthy adults by regulating intracellular zinc concentration through it's interaction with ZnT-1 (3, 4). The role of EVER1 in this complex is unknown, but it is likely that it aids in modulating ZnT-1 activity. This complex works to reduce cytoplasmic and nucleic concentrations of zinc and increase concentration in the ER lumen (3). A mutation in EVER proteins can cause an increase in cytoplasmic and nucleic zinc concentrations, which allows HPV to replicate in the cell, leading to an increased susceptibility to HPV infection. It is also proposed that TMC6 and TMC8 induces tumor necrosis factor-alpha and tumor necrosis factor-related apoptosis-inducing ligand dependent apoptosis (4). This pathway is "critical for preventing the persistence of HPV viruses within cells" (4).
|
References:
1. Epidermodysplasia verruciformis. (n.d.). Retrieved from https://rarediseases.info.nih.gov/diseases/6357/epidermodysplasia-verruciformis
2. TMC6 gene - Genetics Home Reference - NIH. (n.d.). Retrieved from https://ghr.nlm.nih.gov/gene/TMC6
3. Pryzbyszewska, J. (2016, December 21). Re-evaluation of epidermodysplasia verruciformis: Reconciling more than 90 years of debate. Retrieved February 8, 2019, from https://www-sciencedirect-com.ezproxy.library.wisc.edu/science/article/pii/S0190962216312968
4. Kalińska-Bienias, A., Kowalewski, C., & Majewski, S. (2016). The EVER genes - the genetic etiology of carcinogenesis in epidermodysplasia verruciformis and a possible role in non-epidermodysplasia verruciformis patients. Postepy dermatologii i alergologii, 33(2), 75-80.
5. Horton, J. S., & Stokes, A. J. (2014). The transmembrane channel-like protein family and human papillomaviruses: Insights into epidermodysplasia verruciformis and progression to squamous cell carcinoma. Oncoimmunology, 3(1), e28288.
6. Treat, J., & Sullivan, K. E. (2014, August 15). Epidermodysplasia Verruciformis. Retrieved from https://www.sciencedirect.com/science/article/pii/B9780124055469000376
Figures:
https://www.thesun.co.uk/news/5453632/bangladesh-tree-man-bark-like-warts-grow-hand-surgery-remove/
https://escholarship.org/uc/item/7069t3v9
https://ghr.nlm.nih.gov/gene/TMC6#location
https://bmcgenomics.biomedcentral.com/articles/10.1186/1471-2164-4-24#Tab1
https://www.cnn.com/2016/02/26/asia/bangladesh-tree-man-surgery/index.html
1. Epidermodysplasia verruciformis. (n.d.). Retrieved from https://rarediseases.info.nih.gov/diseases/6357/epidermodysplasia-verruciformis
2. TMC6 gene - Genetics Home Reference - NIH. (n.d.). Retrieved from https://ghr.nlm.nih.gov/gene/TMC6
3. Pryzbyszewska, J. (2016, December 21). Re-evaluation of epidermodysplasia verruciformis: Reconciling more than 90 years of debate. Retrieved February 8, 2019, from https://www-sciencedirect-com.ezproxy.library.wisc.edu/science/article/pii/S0190962216312968
4. Kalińska-Bienias, A., Kowalewski, C., & Majewski, S. (2016). The EVER genes - the genetic etiology of carcinogenesis in epidermodysplasia verruciformis and a possible role in non-epidermodysplasia verruciformis patients. Postepy dermatologii i alergologii, 33(2), 75-80.
5. Horton, J. S., & Stokes, A. J. (2014). The transmembrane channel-like protein family and human papillomaviruses: Insights into epidermodysplasia verruciformis and progression to squamous cell carcinoma. Oncoimmunology, 3(1), e28288.
6. Treat, J., & Sullivan, K. E. (2014, August 15). Epidermodysplasia Verruciformis. Retrieved from https://www.sciencedirect.com/science/article/pii/B9780124055469000376
Figures:
https://www.thesun.co.uk/news/5453632/bangladesh-tree-man-bark-like-warts-grow-hand-surgery-remove/
https://escholarship.org/uc/item/7069t3v9
https://ghr.nlm.nih.gov/gene/TMC6#location
https://bmcgenomics.biomedcentral.com/articles/10.1186/1471-2164-4-24#Tab1
https://www.cnn.com/2016/02/26/asia/bangladesh-tree-man-surgery/index.html